DSS-induced damage to basement membranes is repaired by matrix replacement and crosslinking
- PMID: 30837285
- PMCID: PMC6467488
- DOI: 10.1242/jcs.226860
DSS-induced damage to basement membranes is repaired by matrix replacement and crosslinking
Abstract
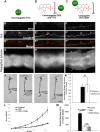
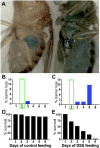
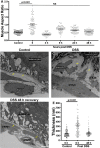
Basement membranes are an ancient form of animal extracellular matrix. As important structural and functional components of tissues, basement membranes are subject to environmental damage and must be repaired while maintaining functions. Little is known about how basement membranes get repaired. This paucity stems from a lack of suitable in vivo models for analyzing such repair. Here, we show that dextran sodium sulfate (DSS) directly damages the gut basement membrane when fed to adult Drosophila DSS becomes incorporated into the basement membrane, promoting its expansion while decreasing its stiffness, which causes morphological changes to the underlying muscles. Remarkably, two days after withdrawal of DSS, the basement membrane is repaired by all measures of analysis. We used this new damage model to determine that repair requires collagen crosslinking and replacement of damaged components. Genetic and biochemical evidence indicates that crosslinking is required to stabilize the newly incorporated repaired Collagen IV rather than to stabilize the damaged Collagen IV. These results suggest that basement membranes are surprisingly dynamic.
Keywords: Basement membrane; Collagen IV; Dextran sodium sulfate; Drosophila; Enterocytes; Matrix stiffness; Midgut.
© 2019. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures





References
-
- Bhave G., Cummings C. F., Vanacore R. M., Kumagai-Cresse C., Ero-Tolliver I. A., Rafi M., Kang J.-S., Pedchenko V., Fessler L. I., Fessler J. H. et al. (2012). Peroxidasin forms sulfilimine chemical bonds using hypohalous acids in tissue genesis. Nat. Chem. Biol. 8, 784-790. 10.1038/nchembio.1038 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R35 GM125028/GM/NIGMS NIH HHS/United States
- T32 HD007502/HD/NICHD NIH HHS/United States
- R21 AR072510/AR/NIAMS NIH HHS/United States
- U2C DK059637/DK/NIDDK NIH HHS/United States
- R01 DK116964/DK/NIDDK NIH HHS/United States
- R25 GM062459/GM/NIGMS NIH HHS/United States
- T32 AI007281/AI/NIAID NIH HHS/United States
- P30 EY008126/EY/NEI NIH HHS/United States
- P30 DK020593/DK/NIDDK NIH HHS/United States
- P30 CA068485/CA/NCI NIH HHS/United States
- R01 DK103831/DK/NIDDK NIH HHS/United States
- U24 DK059637/DK/NIDDK NIH HHS/United States
- P30 DK058404/DK/NIDDK NIH HHS/United States
- F31 HL136081/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

