Determinants of Raft Partitioning of the Helicobacter pylori Pore-Forming Toxin VacA
- PMID: 29531133
- PMCID: PMC5913846
- DOI: 10.1128/IAI.00872-17
Determinants of Raft Partitioning of the Helicobacter pylori Pore-Forming Toxin VacA
Abstract
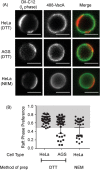
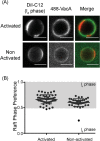
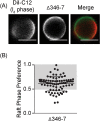
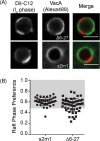
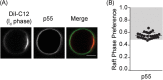
Helicobacter pylori, a Gram-negative bacterium, is a well-known risk factor for gastric cancer. H. pylori vacuolating cytotoxin A (VacA) is a secreted pore-forming toxin that induces a wide range of cellular responses. Like many other bacterial toxins, VacA has been hypothesized to utilize lipid rafts to gain entry into host cells. Here, we used giant plasma membrane vesicles (GPMVs) as a model system to understand the preferential partitioning of VacA into lipid rafts. We show that a wild-type (WT) toxin predominantly associates with the raft phase. Acid activation of VacA enhances binding of the toxin to GPMVs but is not required for raft partitioning. VacA mutant proteins with alterations at the amino terminus (resulting in impaired membrane channel formation) and a nonoligomerizing VacA mutant protein retain the ability to preferentially associate with lipid rafts. Consistent with these results, the isolated VacA p55 domain was capable of binding to lipid rafts. We conclude that the affinity of VacA for rafts is independent of its capacity to oligomerize or form membrane channels.
Keywords: GPMV; Helicobacter pylori; VacA; lipid rafts; oligomerization; ordered phase; phase separation; pore formation; pore-forming toxins.
Copyright © 2018 American Society for Microbiology.
Figures

References
-
- Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. 2013. GLOBOCAN 2012 v1.0, cancer incidence and mortality worldwide: IARC CancerBase. No. 11. International Agency for Research on Cancer, Lyon, France. http://globocan.iarc.fr.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

