Decidual stromal cell-derived PGE2 regulates macrophage responses to microbial threat
- PMID: 30084522
- PMCID: PMC6461368
- DOI: 10.1111/aji.13032
Decidual stromal cell-derived PGE2 regulates macrophage responses to microbial threat
Abstract
Problem: Bacterial chorioamnionitis causes adverse pregnancy outcomes, yet host-microbial interactions are not well characterized within gestational membranes. The decidua, the outermost region of the membranes, is a potential point of entry for bacteria ascending from the vagina to cause chorioamnionitis. We sought to determine whether paracrine communication between decidual stromal cells and macrophages shaped immune responses to microbial sensing.
Method of study: Decidual cell-macrophage interactions were modeled in vitro utilizing decidualized, telomerase-immortalized human endometrial stromal cells (dTHESCs) and phorbol ester-differentiated THP-1 macrophage-like cells. The production of inflammatory mediators in response to LPS was monitored by ELISA for both cell types, while phagocytosis of bacterial pathogens (Escherichia coli and Group B Streptococcus (GBS)) was measured in THP-1 cells or primary human placental macrophages. Diclofenac, a non-selective cyclooxygenase inhibitor, and prostaglandin E2 (PGE2 ) were utilized to interrogate prostaglandins as decidual cell-derived paracrine immunomodulators. A mouse model of ascending chorioamnionitis caused by GBS was utilized to assess the colocalization of bacteria and macrophages in vivo and assess PGE2 production.
Results: In response to LPS, dTHESC and THP-1 coculture demonstrated enhancement of most inflammatory mediators, but a potent suppression of macrophage TNF-α generation was observed. This appeared to reflect a paracrine-mediated effect of decidual cell-derived PGE2 . In mice with GBS chorioamnionitis, macrophages accumulated at sites of bacterial invasion with increased PGE2 in amniotic fluid, suggesting such paracrine effects might hold relevance in vivo.
Conclusion: These data suggest key roles for decidual stromal cells in modulating tissue responses to microbial threat through release of PGE2 .
Keywords: chorioamnionitis; fetal membranes; infection; microfluidics; pregnancy; prostaglandins.
© 2018 John Wiley & Sons A/S. Published by John Wiley & Sons Ltd.
Figures
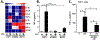

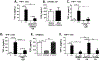



References
-
- Meinert M, Malmström A, Petersen AC, Eriksen GV, Uldbjerg N: Chorioamniontis in preterm delivery is associated with degradation of decorin and biglycan and depletion of hyaluronan in fetal membranes. Placenta;35:546–551. - PubMed
-
- Qazi SA, Stoll BJ: Neonatal sepsis: a major global public health challenge. Pediatr Infect Dis J 2009;28:S1–2. - PubMed
-
- Ofman G, Vasco N, Cantey JB: Risk of Early-Onset Sepsis following Preterm, Prolonged Rupture of Membranes with or without Chorioamnionitis. Am J Perinatol 2016;33:339–342. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- Translational Pathology Shared Resource/International
- P30 CA016087/CA/NCI NIH HHS/United States
- T32 AI007474/AI/NIAID NIH HHS/United States
- Global Alliance to Prevent Prematurity and Stillbirth/International
- 6-FY17-295/March of Dimes/International
- S10 OD018338/OD/NIH HHS/United States
- U2C DK059637/DK/NIDDK NIH HHS/United States
- T32 HD060554/HD/NICHD NIH HHS/United States
- R25 GM062459/GM/NIGMS NIH HHS/United States
- 83573601/U.S. EPA/International
- R01 AI134036/AI/NIAID NIH HHS/United States
- P30 CA068485/CA/NCI NIH HHS/United States
- S10 OD010584/OD/NIH HHS/United States
- T32 HD068256/HD/NICHD NIH HHS/United States
- U24 DK059637/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

