Selective inhibition of mTORC1 in tumor vessels increases antitumor immunity
- PMID: 32759497
- PMCID: PMC7455083
- DOI: 10.1172/jci.insight.139237
Selective inhibition of mTORC1 in tumor vessels increases antitumor immunity
Abstract
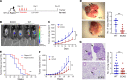
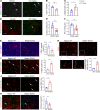
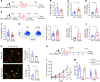
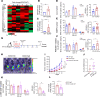
A tumor blood vessel is a key regulator of tissue perfusion, immune cell trafficking, cancer metastasis, and therapeutic responsiveness. mTORC1 is a signaling node downstream of multiple angiogenic factors in the endothelium. However, mTORC1 inhibitors have limited efficacy in most solid tumors, in part due to inhibition of immune function at high doses used in oncology patients and compensatory PI3K signaling triggered by mTORC1 inhibition in tumor cells. Here we show that low-dose RAD001/everolimus, an mTORC1 inhibitor, selectively targets mTORC1 signaling in endothelial cells (ECs) without affecting tumor cells or immune cells, resulting in tumor vessel normalization and increased antitumor immunity. Notably, this phenotype was recapitulated upon targeted inducible gene ablation of the mTORC1 component Raptor in tumor ECs (RaptorECKO). Tumors grown in RaptorECKO mice displayed a robust increase in tumor-infiltrating lymphocytes due to GM-CSF-mediated activation of CD103+ dendritic cells and displayed decreased tumor growth and metastasis. GM-CSF neutralization restored tumor growth and metastasis, as did T cell depletion. Importantly, analyses of human tumor data sets support our animal studies. Collectively, these findings demonstrate that endothelial mTORC1 is an actionable target for tumor vessel normalization, which could be leveraged to enhance antitumor immune therapies.
Keywords: Cancer immunotherapy; Immunology; Oncology; endothelial cells.
Conflict of interest statement
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

