Profile of Podocyte Translatome During Development of Type 2 and Type 1 Diabetic Nephropathy Using Podocyte-Specific TRAP mRNA RNA-seq
- PMID: 34233930
- PMCID: PMC8576501
- DOI: 10.2337/db21-0110
Profile of Podocyte Translatome During Development of Type 2 and Type 1 Diabetic Nephropathy Using Podocyte-Specific TRAP mRNA RNA-seq
Abstract
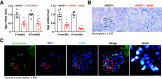
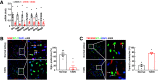
Podocyte injury is important in development of diabetic nephropathy (DN). Although several studies have reported single-cell-based RNA sequencing (RNA-seq) of podocytes in type 1 DN (T1DN), the podocyte translating mRNA profile in type 2 DN (T2DN) has not previously been compared with that of T1DN. We analyzed the podocyte translatome in T2DN in podocin-Cre; Rosa26fsTRAP; eNOS-/-; db/db mice and compared it with that of streptozotocin-induced T1DN in podocin-Cre; Rosa26fsTRAP; eNOS-/- mice using translating ribosome affinity purification (TRAP) and RNA-seq. More than 125 genes were highly enriched in the podocyte ribosome. More podocyte TRAP genes were differentially expressed in T2DN than in T1DN. TGF-β signaling pathway genes were upregulated, while MAPK pathway genes were downregulated only in T2DN, while ATP binding and cAMP-mediated signaling genes were downregulated only in T1DN. Genes regulating actin filament organization and apoptosis increased, while genes regulating VEGFR signaling and glomerular basement membrane components decreased in both type 1 and type 2 diabetic podocytes. A number of diabetes-induced genes not previously linked to podocyte injury were confirmed in both mouse and human DN. On the basis of differences and similarities in the podocyte translatome in T2DN and T1DN, investigators can identify factors underlying the pathophysiology of DN and novel therapeutic targets to treat diabetes-induced podocyte injury.
© 2021 by the American Diabetes Association.
Figures
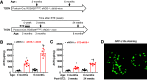

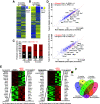
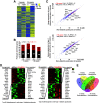
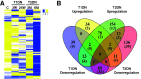
References
-
- Dronavalli S, Duka I, Bakris GL. The pathogenesis of diabetic nephropathy. Nat Clin Pract Endocrinol Metab 2008;4:444–452 - PubMed
-
- Li JJ, Kwak SJ, Jung DS, et al. Podocyte biology in diabetic nephropathy. Kidney Int Suppl 2007. 106:S36–S42 - PubMed
-
- Reddy GR, Kotlyarevska K, Ransom RF, Menon RK. The podocyte and diabetes mellitus: is the podocyte the key to the origins of diabetic nephropathy? Curr Opin Nephrol Hypertens 2008;17:32–36 - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

