Exposure to p40 in Early Life Prevents Intestinal Inflammation in Adulthood Through Inducing a Long-Lasting Epigenetic Imprint on TGFβ
- PMID: 33482393
- PMCID: PMC8020481
- DOI: 10.1016/j.jcmgh.2021.01.004
Exposure to p40 in Early Life Prevents Intestinal Inflammation in Adulthood Through Inducing a Long-Lasting Epigenetic Imprint on TGFβ
Abstract
Background & aims: Colonization by gut microbiota in early life confers beneficial effects on immunity throughout the host's lifespan. We sought to elucidate the mechanisms whereby neonatal supplementation with p40, a probiotic functional factor, reprograms intestinal epithelial cells for protection against adult-onset intestinal inflammation.
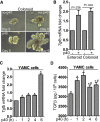
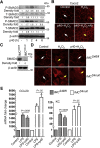
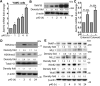
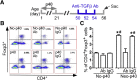
Methods: p40 was used to treat young adult mouse colonic (YAMC) epithelial cells with and without deletion of a methyltransferase, su(var)3-9, enhancer-of-zeste and trithorax domain-containing 1β (Setd1β), and mice in early life or in adulthood. Anti-transforming growth factor β (TGFβ)-neutralizing antibodies were administered to adult mice with and without colitis induced by 2,4,6-trinitrobenzenesulfonic acid or dextran sulfate sodium. We examined Setd1b and Tgfb gene expression, TGFβ production, monomethylation and trimethylation of histone H3 on the lysine 4 residue (H3K4me1/3), H3K4me3 enrichment in Tgfb promoter, differentiation of regulatory T cells (Tregs), and the inflammatory status.
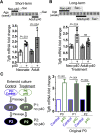
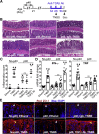
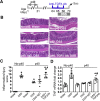
Results: p40 up-regulated expression of Setd1b in YAMC cells. Accordingly, p40 enhanced H3K4me1/3 in YAMC cells in a Setd1β-dependent manner. p40-regulated Setd1β mediated programming the TGFβ locus into a transcriptionally permissive chromatin state and promoting TGFβ production in YAMC. Furthermore, transient exposure to p40 during the neonatal period and in adulthood resulted in the immediate increase in Tgfb gene expression. However, only neonatal p40 supplementation induced the sustained H3K4me1/3 and Tgfb gene expression that persisted into adulthood. Interfering with TGFβ function by neutralizing antibodies diminished the long-lasting effects of neonatal p40 supplementation on differentiation of Tregs and protection against colitis in adult mice.
Conclusions: Exposure to p40 in early life enables an epigenetic imprint on TGFβ, leading to long-lasting production of TGFβ by intestinal epithelial cells to expand Tregs and protect the gut against inflammation.
Keywords: Histone Methyltransferase; Intestinal Epithelial Cell; Probiotic Function Factor; Regulatory T Cell.
Copyright © 2021 The Authors. Published by Elsevier Inc. All rights reserved.
Figures




References
-
- Lilly D.M., Stillwell R.H. Probiotics: growth-promoting factors produced by microorganisms. Science. 1965;147:747–748. - PubMed
-
- Hill C., Guarner F., Reid G., Gibson G.R., Merenstein D.J., Pot B., Morelli L., Canani R.B., Flint H.J., Salminen S., Calder P.C., Sanders M.E. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11:506–514. - PubMed
-
- Vanderpool C., Yan F., Polk D.B. Mechanisms of probiotic action: implications for therapeutic applications in inflammatory bowel diseases. Inflamm Bowel Dis. 2008;14:1585–1596. - PubMed
-
- Lichtenstein L., Avni-Biron I., Ben-Bassat O. Probiotics and prebiotics in Crohn's disease therapies. Best Pract Res Clin Gastroenterol. 2016;30:81–88. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

