A human milk oligosaccharide prevents intestinal inflammation in adulthood via modulating gut microbial metabolism
- PMID: 38441000
- PMCID: PMC11005405
- DOI: 10.1128/mbio.00298-24
A human milk oligosaccharide prevents intestinal inflammation in adulthood via modulating gut microbial metabolism
Abstract
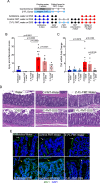
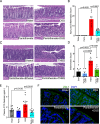
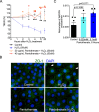
Observational evidence suggests that human milk oligosaccharides (HMOs) promote the growth of commensal bacteria in early life and adulthood. However, the mechanisms by which HMOs benefit health through modulation of gut microbial homeostasis remain largely unknown. 2'-fucosyllactose (2'-FL) is the most abundant oligosaccharide in human milk and contributes to the essential health benefits associated with human milk consumption. Here, we investigated how 2'-FL prevents colitis in adulthood through its effects on the gut microbial community. We found that the gut microbiota from adult mice that consumed 2'-FL exhibited an increase in abundance of several health-associated genera, including Bifidobacterium and Lactobacillus. The 2'-FL-modulated gut microbial community exerted preventive effects on colitis in adult mice. By using Bifidobacterium infantis as a 2'-FL-consuming bacterial model, exploratory metabolomics revealed novel 2'-FL-enriched secretory metabolites by Bifidobacterium infantis, including pantothenol. Importantly, pantothenate significantly protected the intestinal barrier against oxidative stress and mitigated colitis in adult mice. Furthermore, microbial metabolic pathway analysis identified 26 dysregulated metabolic pathways in fecal microbiota from patients with ulcerative colitis, which were significantly regulated by 2'-FL treatment in adult mice, indicating that 2'-FL has the potential to rectify dysregulated microbial metabolism in colitis. These findings support the contribution of the 2'-FL-shaped gut microbial community and bacterial metabolite production to the protection of intestinal integrity and prevention of intestinal inflammation in adulthood.IMPORTANCEAt present, neither basic research nor clinical studies have revealed the exact biological functions or mechanisms of action of individual oligosaccharides during development or in adulthood. Thus, it remains largely unknown whether human milk oligosaccharides could serve as effective therapeutics for gastrointestinal-related diseases. Results from the present study uncover 2'-FL-driven alterations in bacterial metabolism and identify novel B. infantis-secreted metabolites following the consumption of 2'-FL, including pantothenol. This work further demonstrates a previously unrecognized role of pantothenate in significantly protecting the intestinal barrier against oxidative stress and mitigating colitis in adult mice. Remarkably, 2'-FL-enhanced bacterial metabolic pathways are found to be dysregulated in the fecal microbiota of ulcerative colitis patients. These novel metabolic pathways underlying the bioactivities of 2'-FL may lay a foundation for applying individual oligosaccharides for prophylactic intervention for diseases associated with impaired intestinal homeostasis.
Keywords: 2′-fucosyllactose; Bifidobacterium longum subsp. infantis; human milk oligosaccharide; inflammatory bowel diseases; intestinal barrier; pantothenate; the gut microbiota.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Duar RM, Casaburi G, Mitchell RD, Scofield LNC, Ortega Ramirez CA, Barile D, Henrick BM, Frese SA. 2020. Comparative genome analysis of Bifidobacterium longum subsp infantis strains reveals variation in human milk oligosaccharide utilization genes among commercial probiotics. Nutrients 12:3247. doi: 10.3390/nu12113247 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

