Pressure-dependent modulation of inward-rectifying K+ channels: implications for cation homeostasis and K+ dynamics in glaucoma
- PMID: 31166711
- PMCID: PMC6732416
- DOI: 10.1152/ajpcell.00444.2018
Pressure-dependent modulation of inward-rectifying K+ channels: implications for cation homeostasis and K+ dynamics in glaucoma
Abstract
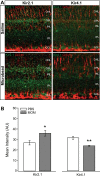
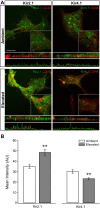
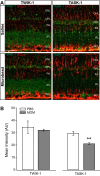
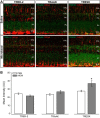
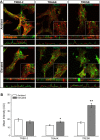
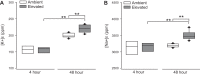
Glaucoma is the leading cause of blindness worldwide, resulting from degeneration of retinal ganglion cells (RGCs), which form the optic nerve. Prior to structural degeneration, RGCs exhibit physiological deficits. Müller glia provide homeostatic regulation of ions that supports RGC physiology through a process called K+ siphoning. Recent studies suggest that several retinal conditions, including glaucoma, involve changes in the expression of K+ channels in Müller glia. To clarify whether glaucoma-related stressors directly alter expression and function of K+ channels in Müller glia, we examined changes in the expression of inwardly rectifying K+ (Kir) channels and two-pore domain (K2P) channels in response to elevated intraocular pressure (IOP) in vivo and in vitro in primary cultures of Müller glia exposed to elevated hydrostatic pressure. We then measured outcomes of cell health, cation homeostasis, and cation flux in Müller glia cultures. Transcriptome analysis in a murine model of microbead-induced glaucoma revealed pressure-dependent downregulation of Kir and K2P channels in vivo. Changes in the expression and localization of Kir and K2P channels in response to elevated pressure were also found in Müller glia in vitro. Finally, we found that elevated pressure compromises the plasma membrane of Müller glia and induces cation dyshomeostasis that involves changes in ion flux through cation channels. Pressure-induced changes in cation flux precede both cation dyshomeostasis and membrane compromise. Our findings have implications for Müller glia responses to pressure-related conditions, i.e., glaucoma, and identify cation dyshomeostasis as a potential contributor to electrophysiological impairment observed in RGCs of glaucomatous retina.
Keywords: K channels; K siphoning; K2P; Kir; Müller glia; glaucoma; microbead; retina.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

