A phase 3 randomized clinical trial using a once-weekly glucagon-like peptide-1 receptor agonist in adolescents and young adults with hypothalamic obesity
- PMID: 33026160
- PMCID: PMC7821019
- DOI: 10.1111/dom.14224
A phase 3 randomized clinical trial using a once-weekly glucagon-like peptide-1 receptor agonist in adolescents and young adults with hypothalamic obesity
Abstract
Aim: To evaluate the efficacy, safety and tolerability of a glucagon-like peptide-1 receptor agonist (GLP-1 RA) in patients with hypothalamic obesity (HO).
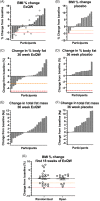
Materials and methods: A two-arm, randomized, multicentre, double-blind, placebo-controlled trial was conducted in 10- to 25-year-olds with hypothalamic injury following intracranial tumour and HO. Participants were randomized to once-weekly subcutaneous injections of a GLP-1 RA exenatide 2 mg (ExQW) or placebo for 36 weeks. The primary efficacy endpoint was 36-week % change in body mass index (BMI). Secondary outcomes included change in body composition (by dual energy x-ray absorptiometry).
Results: Forty-two participants were randomized to ExQW (n = 23) or placebo (n = 19). Participants were 5 ± 2 years (mean ± SD) postdiagnosis and development of HO (BMI 37.3 ± 7.1 kg/m2 ). In intention-to-treat analysis, the effect of 36-week ExQW vs. placebo on % Δ BMI was not significant (estimated treatment difference -1.7 ± 1.8%, 95% CI -4.1 to 0.6%, P = .40); however, total body fat mass was reduced (estimated treatment difference -3.1 ± 1.4 kg, 95% CI -5.7 to -0.4 kg, P = .02). There was a significant reduction in waist circumference (estimated effect of treatment -3.5 [95% CI -5.5 to -1.6] cm, P = .004). All patients treated with placebo increased % of adipose tissue, while 50% treated with ExQW had reductions (P < .001). Mean HbA1c, glucose tolerance and serum lipids did not change significantly with therapy. ExQW was well tolerated. The most frequent adverse events were transient gastrointestinal disturbances (ExQW vs. placebo: nausea 6/23 vs. 3/18, vomiting 4/23 vs. 4/18 and diarrhoea 7/23 vs. 3/18).
Conclusions: GLP-1 RAs are a promising and safe treatment to improve or stabilize HO in children and young adults.
Keywords: antiobesity drug; exenatide; randomized trial.
© 2020 The Authors. Diabetes, Obesity and Metabolism published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Muller HL. Childhood craniopharyngioma. Recent advances in diagnosis, treatment and follow‐up. Horm Res. 2008;69(4):193‐202. - PubMed
-
- Simoneau‐Roy J, O'Gorman C, Pencharz P, Adeli K, Daneman D, Hamilton J. Insulin sensitivity and secretion in children and adolescents with hypothalamic obesity following treatment for craniopharyngioma. Clin Endocrinol. 2010;72(3):364‐370. - PubMed
-
- Bereket A, Kiess W, Lustig RH, et al. Hypothalamic obesity in children. Obes Rev. 2012;13(9):780‐798. - PubMed
-
- Elowe‐Gruau E, Beltrand J, Brauner R, et al. Childhood craniopharyngioma: hypothalamus‐sparing surgery decreases the risk of obesity. J Clin Endocrinol Metab. 2013;98(6):2376‐2382. - PubMed
-
- Muller HL, Gebhardt U, Teske C, et al. Post‐operative hypothalamic lesions and obesity in childhood craniopharyngioma: results of the multinational prospective trial KRANIOPHARYNGEOM 2000 after 3‐year follow‐up. Eur J Endocrinol. 2011;165(1):17‐24. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

