INTRAPULMONARY TREATMENT WITH A NOVEL TLR4 AGONIST CONFERS PROTECTION AGAINST KLEBSIELLA PNEUMONIA
- PMID: 36018281
- PMCID: PMC9647733
- DOI: 10.1097/SHK.0000000000001977
INTRAPULMONARY TREATMENT WITH A NOVEL TLR4 AGONIST CONFERS PROTECTION AGAINST KLEBSIELLA PNEUMONIA
Abstract
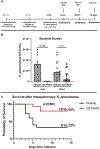
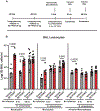
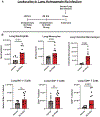
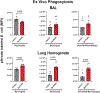
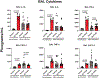
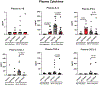
Objectives: Nosocomial pneumonia is a common complication in critically ill patients. The goal of this study was to examine the efficacy of the Toll-like receptor 4 agonist 3-deacyl phosphorylated hexacyl disaccharide (3D PHAD), in a clinically relevant murine model of pneumonia, and assess the cellular mechanisms that mediate the protective response. Design: Mice received intrapulmonary 3D PHAD (20 μg) or vehicle for 2 consecutive days before challenge with intrapulmonary Klebsiella pneumoniae (2.3 × 10 3 colony-forming units). Mice were followed for 14-day survival, pulmonary K. pneumoniae burden, lung leukocyte profile, leukocyte phagocytic capacity, and cytokine production. Pneumonia severity and leukocyte recruitment were further assessed by histological evaluation. Setting: Research laboratory. Subjects: Wild-type, male C57BL/6 J mice. Interventions: Intrapulmonary treatment with 20 μg 3D PHAD for 2 consecutive days. Measurements and main results: Intrapulmonary treatment with 3D PHAD decreased lung K. pneumoniae colony-forming units and pneumonia severity with an associated improvement in survival compared with mice treated with vehicle. The numbers of neutrophils, monocytes, and macrophages in the lungs of 3D PHAD-treated mice were higher than those in vehicle-treated mice before infection but were not significantly different from vehicle-treated mice at 48 h after K. pneumoniae challenge. Lung innate leukocytes from 3D PHAD-treated mice had increased phagocytic capacity. Treatment with 3D PHAD alone increased cytokines in the lungs but decreased cytokines in plasma during K. pneumoniae pneumonia as compared with control. Conclusions: Intrapulmonary treatment with 3D PHAD augments innate immunity in the lung and facilitates resistance to K. pneumoniae pneumonia.
Copyright © 2022 by the Shock Society.
Conflict of interest statement
The authors report no conflicts of interest.
Figures
References
-
- Forrester JD, Maggio PM, Tennakoon L: Cost of health care–associated infections in the United States. J Patient Saf 18(2):e477–e479, 2022. - PubMed
-
- Torres A, Niederman MS, Chastre J, Ewig S, Fernandez-Vandellos P, Hanberger H, Kollef M, Bassi GL, Luna CM, Martin-Loeches I, et al. : International ERS/ESICM/ESCMID/ALAT guidelines for the management of hospital-acquired pneumonia and ventilator-associated pneumonia: guidelines for the management of hospital-acquired pneumonia (HAP)/ventilator-associated pneumonia (VAP) of the European Respiratory Society (ERS), European Society of Intensive Care Medicine (ESICM), European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and Asociacion Latinoamericana del Torax (ALAT). Eur Respir J 50(3):1700582, 2017. - PubMed
-
- Hampton T: Novel programs and discoveries aim to combat antibiotic resistance. JAMA 313(24):2411–2413, 2015. - PubMed
-
- Marston HD, Dixon DM, Knisely JM, Palmore TN, Fauci AS: Antimicrobial resistance. JAMA 316(11):1193–1204, 2016. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

