GABAA receptor β3 subunit mutation D120N causes Lennox-Gastaut syndrome in knock-in mice
- PMID: 32467926
- PMCID: PMC7238755
- DOI: 10.1093/braincomms/fcaa028
GABAA receptor β3 subunit mutation D120N causes Lennox-Gastaut syndrome in knock-in mice
Abstract
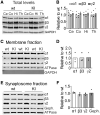
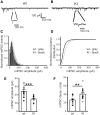
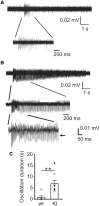
The Lennox-Gastaut syndrome is a devastating early-onset epileptic encephalopathy, associated with severe behavioural abnormalities. Its pathophysiology, however, is largely unknown. A de novo mutation (c.G358A, p.D120N) in the human GABA type-A receptor β3 subunit gene (GABRB3) has been identified in a patient with Lennox-Gastaut syndrome. To determine whether the mutation causes Lennox-Gastaut syndrome in vivo in mice and to elucidate its mechanistic effects, we generated the heterozygous Gabrb3+/D120N knock-in mouse and found that it had frequent spontaneous atypical absence seizures, as well as less frequent tonic, myoclonic, atonic and generalized tonic-clonic seizures. Each of these seizure types had a unique and characteristic ictal EEG. In addition, knock-in mice displayed abnormal behaviours seen in patients with Lennox-Gastaut syndrome including impaired learning and memory, hyperactivity, impaired social interactions and increased anxiety. This Gabrb3 mutation did not alter GABA type-A receptor trafficking or expression in knock-in mice. However, cortical neurons in thalamocortical slices from knock-in mice had reduced miniature inhibitory post-synaptic current amplitude and prolonged spontaneous thalamocortical oscillations. Thus, the Gabrb3+/D120N knock-in mouse recapitulated human Lennox-Gastaut syndrome seizure types and behavioural abnormalities and was caused by impaired inhibitory GABAergic signalling in the thalamocortical loop. In addition, treatment with antiepileptic drugs and cannabinoids ameliorated atypical absence seizures in knock-in mice. This congenic knock-in mouse demonstrates that a single-point mutation in a single gene can cause development of multiple types of seizures and multiple behavioural abnormalities. The knock-in mouse will be useful for further investigation of the mechanisms of Lennox-Gastaut syndrome development and for the development of new antiepileptic drugs and treatments.
Keywords: GABRB3; Lennox–Gastaut syndrome; early-onset epileptic encephalopathy; genetic epilepsies; seizures.
© The Author(s) (2020). Published by Oxford University Press on behalf of the Guarantors of Brain.
Figures




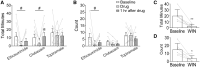
References
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
