Aging compromises human islet beta cell function and identity by decreasing transcription factor activity and inducing ER stress
- PMID: 36197983
- PMCID: PMC9534504
- DOI: 10.1126/sciadv.abo3932
Aging compromises human islet beta cell function and identity by decreasing transcription factor activity and inducing ER stress
Abstract
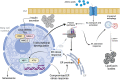
Pancreatic islet beta cells are essential for maintaining glucose homeostasis. To understand the impact of aging on beta cells, we performed meta-analysis of single-cell RNA sequencing datasets, transcription factor (TF) regulon analysis, high-resolution confocal microscopy, and measured insulin secretion from nondiabetic donors spanning most of the human life span. This revealed the range of molecular and functional changes that occur during beta cell aging, including the transcriptional deregulation that associates with cellular immaturity and reorganization of beta cell TF networks, increased gene transcription rates, and reduced glucose-stimulated insulin release. These alterations associate with activation of endoplasmic reticulum (ER) stress and autophagy pathways. We propose that a chronic state of ER stress undermines old beta cell structure function to increase the risk of beta cell failure and type 2 diabetes onset as humans age.
Figures






References
-
- Basu R., Breda E., Oberg A. L., Powell C. C., Man C. D., Basu A., Vittone J. L., Klee G. G., Arora P., Jensen M. D., Toffolo G., Cobelli C., Rizza R. A., Mechanisms of the age-associated deterioration in glucose tolerance: Contribution of alterations in insulin secretion, action, and clearance. Diabetes 52, 1738–1748 (2003). - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

