Plasma Cell Differentiation, Antibody Quality, and Initial Germinal Center B Cell Population Depend on Glucose Influx Rate
- PMID: 37955416
- PMCID: PMC10841396
- DOI: 10.4049/jimmunol.2200756
Plasma Cell Differentiation, Antibody Quality, and Initial Germinal Center B Cell Population Depend on Glucose Influx Rate
Abstract
Serum Ab concentrations, selection for higher affinity BCRs, and generation of higher Ab affinities are important elements of immune response optimization and functions of germinal center (GC) reactions. B cell proliferation requires nutrients to support the anabolism inherent in clonal expansion. Glucose usage by mouse GC B cells has been reported to contribute little to their energy needs, with questions raised as to whether glucose uptake or glycolysis increases in GC B cells compared with their naive precursors. Indeed, metabolism can be highly flexible, such that supply shortage along one pathway may be compensated by increased flux on others. We now show that reduction of the glucose transporter GLUT1 in mice after establishment of a preimmune B cell repertoire, even after initiation of the GC B cell gene expression program, decreased initial GC B cell population numbers, affinity maturation, and plasma cell outputs. Glucose oxidation was heightened in GC B cells, but this hexose flowed more into the pentose phosphate pathway, whose activity was important in controlling reactive oxygen species (ROS) and Ab-secreting cell production. In modeling how glucose usage by B cells promotes the Ab response, the control of ROS appeared insufficient. Surprisingly, the combination of galactose, which mitigated ROS, with provision of mannose, an efficient precursor to glycosylation, supported robust production of and normal Ab secretion by Ab-secreting cells under glucose-free conditions. Collectively, the findings indicate that GCs depend on normal glucose influx, especially in plasma cell production, but reveal an unexpected metabolic flexibility in hexose requirements.
Copyright © 2023 by The American Association of Immunologists, Inc.
Figures

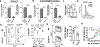
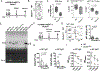

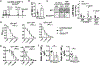

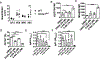
Update of
-
Plasma cell differentiation, antibody quality, and initial germinal center B cell population depend on glucose influx rate.bioRxiv [Preprint]. 2023 Sep 16:2023.09.13.557599. doi: 10.1101/2023.09.13.557599. bioRxiv. 2023. Update in: J Immunol. 2024 Jan 1;212(1):43-56. doi: 10.4049/jimmunol.2200756. PMID: 37745429 Free PMC article. Updated. Preprint.
References
-
- Ripperger TJ and Bhattacharya D. 2021. Transcriptional and metabolic control of memory B cells and plasma cells. Annu Rev Immunol. 39: 345–368. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

