Salmonella enterica serovar Typhimurium uses anaerobic respiration to overcome propionate-mediated colonization resistance
- PMID: 34986344
- PMCID: PMC8800556
- DOI: 10.1016/j.celrep.2021.110180
Salmonella enterica serovar Typhimurium uses anaerobic respiration to overcome propionate-mediated colonization resistance
Abstract
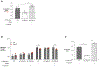
The gut microbiota benefits the host by limiting enteric pathogen expansion (colonization resistance), partially via the production of inhibitory metabolites. Propionate, a short-chain fatty acid produced by microbiota members, is proposed to mediate colonization resistance against Salmonella enterica serovar Typhimurium (S. Tm). Here, we show that S. Tm overcomes the inhibitory effects of propionate by using it as a carbon source for anaerobic respiration. We determine that propionate metabolism provides an inflammation-dependent colonization advantage to S. Tm during infection. Such benefit is abolished in the intestinal lumen of Salmonella-infected germ-free mice. Interestingly, S. Tm propionate-mediated intestinal expansion is restored when germ-free mice are monocolonized with Bacteroides thetaiotaomicron (B. theta), a prominent propionate producer in the gut, but not when mice are monocolonized with a propionate-production-deficient B. theta strain. Taken together, our results reveal a strategy used by S. Tm to mitigate colonization resistance by metabolizing microbiota-derived propionate.
Keywords: Salmonella; colonization resistance; gut microbiota; intestinal inflammation; microbial metabolism; propionate.
Copyright © 2021 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures
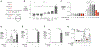
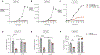



References
-
- Alam MS, Akaike T, Okamoto S, Kubota T, Yoshitake J, Sawa T, Miyamoto Y, Tamura F, and Maeda H (2002). Role of nitric oxide in host defense in murine salmonellosis as a function of its antibacterial and antiapoptotic activities. Infect. Immun 70, 3130–3142. 10.1128/iai.70.6.3130-3142.2002. - DOI - PMC - PubMed
-
- Barthel M, Hapfelmeier S, Quintanilla-Martínez L, Kremer M, Rohde M, Hogardt M, Pfeffer K, Rüssmann H, and Hardt WD (2003). Pretreatment of mice with streptomycin provides a Salmonella enterica serovar Typhimurium colitis model that allows analysis of both pathogen and host. Infect. Immun 71, 2839–2858. 10.1128/iai.71.5.2839-2858.2003. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

