Nf2 fine-tunes proliferation and tissue alignment during closure of the optic fissure in the embryonic mouse eye
- PMID: 33075808
- PMCID: PMC7749708
- DOI: 10.1093/hmg/ddaa228
Nf2 fine-tunes proliferation and tissue alignment during closure of the optic fissure in the embryonic mouse eye
Abstract
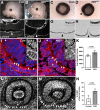
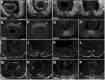
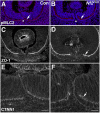
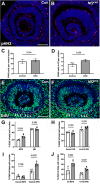
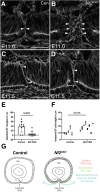
Uveal coloboma represents one of the most common congenital ocular malformations accounting for up to 10% of childhood blindness (~1 in 5000 live birth). Coloboma originates from defective fusion of the optic fissure (OF), a transient gap that forms during eye morphogenesis by asymmetric, ventral invagination. Genetic heterogeneity combined with the activity of developmentally regulated genes suggests multiple mechanisms regulating OF closure. The tumor suppressor and FERM domain protein Neurofibromin 2 (NF2) controls diverse processes in cancer, development and regeneration, via Hippo pathway and cytoskeleton regulation. In humans, NF2 mutations can cause ocular abnormalities, including coloboma, however, its actual role in OF closure is unknown. Using conditional inactivation in the embryonic mouse eye, our data indicate that loss of Nf2 function results in a novel underlying cause for coloboma. In particular, mutant eyes show substantially increased retinal pigmented epithelium (RPE) proliferation in the fissure region with concomitant acquisition of RPE cell fate. Cells lining the OF margin can maintain RPE fate ectopically and fail to transition from neuroepithelial to cuboidal shape. In the dorsal RPE of the optic cup, Nf2 inactivation leads to a robust increase in cell number, with local disorganization of the cytoskeleton components F-actin and pMLC2. We propose that RPE hyperproliferation is the primary cause for the observed defects causing insufficient alignment of the OF margins in Nf2 mutants and failure to fuse properly, resulting in persistent coloboma. Our findings indicate that limiting proliferation particularly in the RPE layer is a critical mechanism during OF closure.
© The Author(s) 2020. Published by Oxford University Press. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures

References
-
- Clementi M., Turolla L., Mammi I. and Tenconi R. (1992) Clinical anophthalmia: an epidemiological study in Northeast Italy based on 368,256 consecutive births. Teratology, 46, 551–553. - PubMed
-
- Williamson K.A. and Fitz Patrick D.R. (2014) The genetic architecture of microphthalmia, anophthalmia and coloboma. Eur. J. Med. Genet., 57, 369–380. - PubMed
-
- Shah S.P., Taylor A.E., Sowden J.C., Ragge N.K., Russell-Eggitt I., Rahi J.S., Gilbert C.E. and Surveillance of Eye Anomalies Special Interest Group (2011) Anophthalmos, microphthalmos, and typical coloboma in the United Kingdom: a prospective study of incidence and risk. Invest. Ophthalmol. Vis. Sci., 52, 558–564. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

