Novel Human NKCC1 Mutations Cause Defects in Goblet Cell Mucus Secretion and Chronic Inflammation
- PMID: 31655271
- PMCID: PMC6957845
- DOI: 10.1016/j.jcmgh.2019.10.006
Novel Human NKCC1 Mutations Cause Defects in Goblet Cell Mucus Secretion and Chronic Inflammation
Abstract
Background & aims: Infections resulting from intestinal yeast and bacteria affect a large number of patients with deficits in absorptive or secretory epithelial transport mechanisms. The basolateral Na+-K+-2Cl- cotransporter (NKCC1) has been implicated in intestinal epithelial fluid secretion. Two patients with deleterious heterozygous (NKCC1-DFX, DFX for Asp-Phe-stop codon) or homozygous (Kilquist) mutations in SLC12A2 (NKCC1) suffered from gastrointestinal deficits. Because of chronic infections, the colon and the small intestine of the NKCC1-DFX patient were resected surgically.
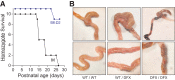
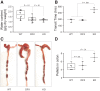
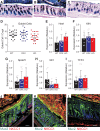
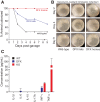
Methods: To investigate how NKCC1 affects the integrity and function of the gut epithelia, we used a mouse model recapitulating the NKCC1-DFX patient mutation. Electron microscopy and immunostaining were used to analyze the integrity of the colonic mucus layers and immune cell infiltration. Fluorescence in situ hybridization was performed on the distal colon sections to measure bacteria translocation to the mucosa and submucosa. Citrobacter rodentium was used to measure mouse ability to clear enteric infection. A multiplex cytokine assay was used to analyze mouse inflammatory response to infection.
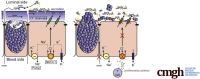
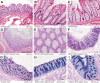
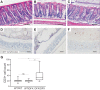
Results: We show that NKCC1-DFX expression causes defective goblet cell mucus granule exocytosis, leading to secretion of intact granules into the lumen of the large intestine. In addition, NKCC1-DFX colon submucosal glands secrete mucus that remained attached to the epithelium. Importantly, expression of the mutant NKCC1 or complete loss of NKCC1 function leads to aggravated inflammatory response to C rodentium infection. Compared with wild-type, NKCC1-DFX mice showed decreased expression of claudin-2, a tight junction protein involved in paracellular Na+ and water transport and enteric infection clearance.
Conclusions: Our data indicate that NKCC1-DFX impairs gut barrier function by affecting mucus secretion and immune properties.
Keywords: Bacterial Infection; Goblet Cell; Mucus Secretion; NKCC1.
Copyright © 2020 The Authors. Published by Elsevier Inc. All rights reserved.
Figures




References
-
- Geck P., Pietrzyk C., Burckhardt B.-C., Pfeiffer B., Heinz E. Electrically silent cotransport of Na+ , K+ and Cl- in Ehrlich cells. Biochim Biophys Acta. 1980;600:432–447. - PubMed
-
- Plotkin M.D., Snyder E.Y., Hebert S.C., Delpire E. Expression of the Na-K-2Cl cotransporter is developmentally regulated in postnatal rat brains: a possible mechanism underlying GABA's excitatory role in immature brain. J Neurobiol. 1997;33:781–795. - PubMed
-
- Dzhala V.I., Talos D.M., Sdrulla D.A., Brumback A.C., Mathews G.C., Benke T.A., Delpire E., Jensen F.E., Staley K.J. NKCC1 transporter facilitates seizures in the developing brain. Nat Med. 2005;11:1205–1213. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

