Modeling intrahippocampal effects of anterior hippocampal hyperactivity relevant to schizophrenia using chemogenetic excitation of long axis-projecting mossy cells in the mouse dentate gyrus
- PMID: 34414387
- PMCID: PMC8372626
- DOI: 10.1016/j.bpsgos.2021.04.005
Modeling intrahippocampal effects of anterior hippocampal hyperactivity relevant to schizophrenia using chemogenetic excitation of long axis-projecting mossy cells in the mouse dentate gyrus
Abstract
Background: The anterior hippocampus of individuals with early psychosis or schizophrenia is hyperactive, as is the ventral hippocampus in many rodent models for schizophrenia risk. Mossy cells (MCs) of the ventral dentate gyrus (DG) densely project in the hippocampal long axis, targeting both dorsal DG granule cells and inhibitory interneurons. Mossy cells are responsive to stimulation throughout hippocampal subfields, and thus may be suited to detect hyperactivity in areas where it originates such as CA1. Here we tested the hypothesis that hyperactivation of ventral MCs activates dorsal DG granule cells to influence dorsal hippocampal function.
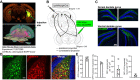
Methods: In CD-1 mice, we targeted dorsal DG-projecting ventral MCs using an adeno-associated virus intersectional strategy. In vivo fiber photometry recording of ventral MCs was performed during exploratory behaviors. We used excitatory chemogenetic constructs to test the effects of ventral MC hyperactivation on long-term spatial memory during an object location memory task.
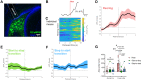
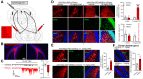
Results: Photometry revealed ventral MCs were activated during exploratory rearing. Ventral MCs made functional monosynaptic inputs to dorsal DG granule cells, and chemogenetic activation of ventral MCs modestly increased activity of dorsal DG granule cells measured by c-Fos. Finally, chemogenetic activation of ventral MCs during the training phase of an object location memory task impaired test performance 24 hours later, without effects on locomotion or object exploration.
Conclusions: These data suggest that ventral MC activation can directly excite dorsal granule cells and interfere with dorsal DG function, supporting future study of their in vivo activity in animal models for schizophrenia featuring ventral hyperactivity.
Keywords: hippocampus; hyperactivity; learning and memory; mossy cells; psychosis; schizophrenia.
Figures


References
-
- Szeszko P.R., Goldberg E., Gunduz-Bruce H., Ashtari M., Robinson D., Malhotra A.K., et al. Smaller anterior hippocampal formation volume in antipsychotic-naive patients with first-episode schizophrenia. Am J Psychiatry. 2003;160:2190–2197. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

