Cytochrome P450 epoxygenase-derived epoxyeicosatrienoic acids contribute to insulin sensitivity in mice and in humans
- PMID: 28352940
- PMCID: PMC5921930
- DOI: 10.1007/s00125-017-4260-0
Cytochrome P450 epoxygenase-derived epoxyeicosatrienoic acids contribute to insulin sensitivity in mice and in humans
Abstract
Aims/hypothesis: Insulin resistance is frequently associated with hypertension and type 2 diabetes. The cytochrome P450 (CYP) arachidonic acid epoxygenases (CYP2C, CYP2J) and their epoxyeicosatrienoic acid (EET) products lower blood pressure and may also improve glucose homeostasis. However, the direct contribution of endogenous EET production on insulin sensitivity has not been previously investigated. In this study, we tested the hypothesis that endogenous CYP2C-derived EETs alter insulin sensitivity by analysing mice lacking CYP2C44, a major EET producing enzyme, and by testing the association of plasma EETs with insulin sensitivity in humans.
Methods: We assessed insulin sensitivity in wild-type (WT) and Cyp2c44 -/- mice using hyperinsulinaemic-euglycaemic clamps and isolated skeletal muscle. Insulin secretory function was assessed using hyperglycaemic clamps and isolated islets. Vascular function was tested in isolated perfused mesenteric vessels. Insulin sensitivity and secretion were assessed in humans using frequently sampled intravenous glucose tolerance tests and plasma EETs were measured by mass spectrometry.
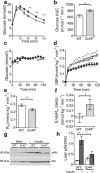
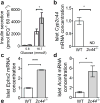
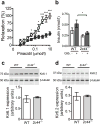
Results: Cyp2c44 -/- mice showed decreased glucose tolerance (639 ± 39.5 vs 808 ± 37.7 mmol/l × min for glucose tolerance tests, p = 0.004) and insulin sensitivity compared with WT controls (hyperinsulinaemic clamp glucose infusion rate average during terminal 30 min 0.22 ± 0.02 vs 0.33 ± 0.01 mmol kg-1 min-1 in WT and Cyp2c44 -/- mice respectively, p = 0.003). Although glucose uptake was diminished in Cyp2c44 -/- mice in vivo (gastrocnemius Rg 16.4 ± 2.0 vs 6.2 ± 1.7 μmol 100 g-1 min-1, p < 0.01) insulin-stimulated glucose uptake was unchanged ex vivo in isolated skeletal muscle. Capillary density was similar but vascular KATP-induced relaxation was impaired in isolated Cyp2c44 -/- vessels (maximal response 39.3 ± 6.5% of control, p < 0.001), suggesting that impaired vascular reactivity produces impaired insulin sensitivity in vivo. Similarly, plasma EETs positively correlated with insulin sensitivity in human participants.
Conclusions/interpretation: CYP2C-derived EETs contribute to insulin sensitivity in mice and in humans. Interventions to increase circulating EETs in humans could provide a novel approach to improve insulin sensitivity and treat hypertension.
Keywords: Arachidonic acid; Epoxygenases; Hypertension; Insulin secretion in vitro and in vivo; Insulin sensitivity.
Conflict of interest statement
The authors declare that there is no duality of interest associated with this manuscript.
Figures
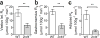
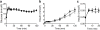


References
-
- Kahn SE, Hull RL, Utzschneider KM. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature. 2006;444:840–846. - PubMed
-
- Reaven G. Insulin resistance and coronary heart disease in nondiabetic individuals. Arterioscler Thromb Vasc Biol. 2012;32:1754–1759. - PubMed
-
- Roman RJ, Maier KG, Sun CW, Harder DR, Alonso-Galicia M. Renal and cardiovascular actions of 20-hydroxyeicosatetraenoic acid and epoxyeicosatrienoic acids. Clin Exp Pharmacol Physiol. 2000;27:855–865. - PubMed
-
- Spector AA, Fang X, Snyder GD, Weintraub NL. Epoxyeicosatrienoic acids (EETs): metabolism and biochemical function. Prog Lipid Res. 2004;43:55–90. - PubMed
MeSH terms
Substances
Grants and funding
- UL1 TR000445/TR/NCATS NIH HHS/United States
- R01 DK096994/DK/NIDDK NIH HHS/United States
- I01 BX002025/BX/BLRD VA/United States
- K23 DK081662/DK/NIDDK NIH HHS/United States
- R01 DK103616/DK/NIDDK NIH HHS/United States
- P30 DK020593/DK/NIDDK NIH HHS/United States
- UL1 RR024975/RR/NCRR NIH HHS/United States
- U24 DK059637/DK/NIDDK NIH HHS/United States
- R01 HL060906/HL/NHLBI NIH HHS/United States
- P01 DK038226/DK/NIDDK NIH HHS/United States
- R01 DK095761/DK/NIDDK NIH HHS/United States
- R01 CA162433/CA/NCI NIH HHS/United States
- K23 DK100533/DK/NIDDK NIH HHS/United States
- P30 DK114809/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

