The age-related decrease in material properties of BALB/c mouse long bones involves alterations to the extracellular matrix
- PMID: 31678497
- PMCID: PMC6885131
- DOI: 10.1016/j.bone.2019.115126
The age-related decrease in material properties of BALB/c mouse long bones involves alterations to the extracellular matrix
Abstract
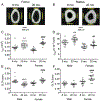
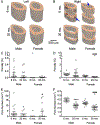
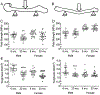
One possibility for the disproportionate increase in fracture risk with aging relative to the decrease in bone mass is an accumulation of changes to the bone matrix which deleteriously affect fracture resistance. In order to effectively develop new targets for osteoporosis, a preclinical model of the age-related loss in fracture resistance needs to be established beyond known age-related decreases in bone mineral density and bone volume fraction. To that end, we examined long bones of male and female BALB/c mice at 6-mo. and 20-mo. of age and assessed whether material and matrix properties of cortical bone significantly differed between the age groups. The second moment of area of the diaphysis (minimum and maximum principals for femur and radius, respectively) as measured by ex vivo micro-computed tomography (μCT) was higher at 20-mo. than at 6-mo. for both males and females, but ultimate moment as measured by three-point bending tests did not decrease with age. Cortical thickness was lower with age for males, but higher for old females. Partially accounting for differences in structure, material estimates of yield, ultimate stress, and toughness (left femur) were 12.6%, 11.1%, and 40.9% lower, respectively, with age for both sexes. The ability of the cortical bone to resist crack growth (right femur) was also 18.1% less for the old than for the young adult mice. These decreases in material properties were not due to changes in intracortical porosity as pore number decreased with age. Rather, age-related alterations in the matrix were observed for both sexes: enzymatic and non-enzymatic crosslinks by high performance liquid chromatography increased (femur), volume fraction of bound water by 1H-nuclear magnetic resonance relaxometry decreased (femur), cortical tissue mineral density by μCT increased (femur and radius), and an Amide I sub-peak ratio I1670/I1640 by Raman spectroscopy increased (tibia). Overall, there are multiple matrix changes to potentially target that could prevent the age-related decrease in fracture resistance observed in BALB/c mouse.
Keywords: Advanced glycation end-products; Bone quality; Bound water; Collagen; Raman spectroscopy; Toughness.
Copyright © 2019 Elsevier Inc. All rights reserved.
Figures


References
-
- Milovanovic P, Adamu U, Simon MJK, Rolvien T, Djuric M, Amling M, et al. , Age- and Sex-Specific Bone Structure Patterns Portend Bone Fragility in Radii and Tibiae in Relation to Osteodensitometry: A High-Resolution Peripheral Quantitative Computed Tomography Study in 385 Individuals, J Gerontol a-Biol. 70 (2015) 1269–1275. doi: 10.1093/gerona/glv052. - DOI - PubMed
-
- Kral R, Osima M, Borgen TT, Vestgaard R, Richardsen E, Bjørnerem Å, Increased cortical porosity and reduced cortical thickness of the proximal femur are associated with nonvertebral fracture independent of Fracture Risk Assessment Tool and Garvan estimates in postmenopausal women, PLoS ONE. 12 (2017) e0185363–15. doi: 10.1371/journal.pone.0185363. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

