Helicobacter pylori- induced cell death is counteracted by NF-κB-mediated transcription of DARPP-32
- PMID: 27590997
- PMCID: PMC5334457
- DOI: 10.1136/gutjnl-2016-312141
Helicobacter pylori- induced cell death is counteracted by NF-κB-mediated transcription of DARPP-32
Abstract
Objective: DARPP-32 is a frequently amplified and overexpressed gene that promotes several oncogenic functions in gastric cancer. Herein, we investigated the relationship between Helicobacter pylori infection, proinflammatory NF-κB activation and regulation of DARPP-32.
Design: The study used in vivo and in vitro experiments. Luciferase reporter, quantitative real-time PCR, immunoblot, chromatin immunoprecipitation (ChIP), cell viability, H. pylori infection, tissue microarrays and immunohistochemical assays were used.
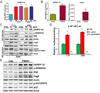
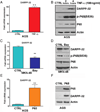
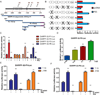
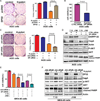
Results: Our results indicated that H. pylori infection increased the DARPP-32 mRNA and protein levels in gastric cancer cell lines and gastric mucosa of mice. H. pylori infection increased the activity of NF-κB reporter and p-NF-κB (S536) protein level in vitro and in vivo. To investigate the transcriptional regulation of DARPP-32, we cloned a 3019 bp of the DARPP-32 promoter into the luciferase reporter (pGL3-Luc). Both H. pylori infection and tumour necrosis factor-α treatment induced DARPP-32 reporter activity (p<0.01). Using deletion constructs of DARPP-32 promoter and ChIP assay, we demonstrated that the sequence -996 to -1008 bp containing putative NF-κB-binding sites is the most active region. The induction of DARPP-32 expression by H. pylori infection counteracted H. pylori-induced cell death through activation of serine/threonine-specific protein kinase (AKT), as determined by ATP-Glo and clonogenic survival assays. Immunohistochemistry analysis demonstrated a significant positive correlation between NF-κB and DARPP-32 expression levels in gastric cancer tissues (r2=0.43, p<0.01).
Conclusions: Given the high frequency of DARPP-32 overexpression and its prosurvival oncogenic functions, the induction of DARPP-32 expression following H. pylori infection and activation of NF-κB provides a link between infection, inflammation and gastric tumourigenesis.
Keywords: GASTRIC CANCER; GASTRIC INFLAMMATION; HELICOBACTER PYLORI.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://www.bmj.com/company/products-services/rights-and-licensing/.
Figures



Comment in
-
Helicobacter pylori: DARPP-32: a link between infection and gastric cancer.Nat Rev Gastroenterol Hepatol. 2016 Nov;13(11):625. doi: 10.1038/nrgastro.2016.159. Epub 2016 Sep 21. Nat Rev Gastroenterol Hepatol. 2016. PMID: 27649788 No abstract available.
-
NF-κB mediated transcription of DARPP-32 prevents Helicobacter pylori-induced cell death.Gut. 2017 May;66(5):761-762. doi: 10.1136/gutjnl-2016-312822. Epub 2016 Oct 27. Gut. 2017. PMID: 27789656 No abstract available.
References
-
- Bertuccio P, Chatenoud L, Levi F, et al. Recent patterns in gastric cancer: a global overview. Int J Cancer. 2009;125:666–673. - PubMed
-
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–E386. - PubMed
-
- El-Rifai W, Smith MF, Jr, Li G, et al. Gastric cancers overexpress DARPP-32 and a novel isoform, t-DARPP. Cancer Res. 2002;62:4061–4064. - PubMed
-
- Belkhiri A, Zaika A, Pidkovka N, et al. Darpp-32: a novel antiapoptotic gene in upper gastrointestinal carcinomas. Cancer Res. 2005;65:6583–6592. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
