Engineering of a Polydisperse Small Heat-Shock Protein Reveals Conserved Motifs of Oligomer Plasticity
- PMID: 29983375
- PMCID: PMC6563326
- DOI: 10.1016/j.str.2018.05.015
Engineering of a Polydisperse Small Heat-Shock Protein Reveals Conserved Motifs of Oligomer Plasticity
Abstract
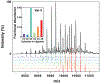
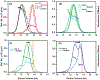
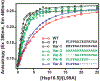
Small heat-shock proteins (sHSPs) are molecular chaperones that bind partially and globally unfolded states of their client proteins. Previously, we discovered that the archaeal Hsp16.5, which forms ordered and symmetric 24-subunit oligomers, can be engineered to transition to an ordered and symmetric 48-subunit oligomer by insertion of a peptide from human HspB1 (Hsp27). Here, we uncovered the existence of an array of oligomeric states (30-38 subunits) that can be populated as a consequence of altering the sequence and length of the inserted peptide. Polydisperse Hsp16.5 oligomers displayed higher affinity to a model client protein consistent with a general mechanism for recognition and binding that involves increased access of the hydrophobic N-terminal region. Our findings, which integrate structural and functional analyses from evolutionarily distant sHSPs, support a model wherein the modular architecture of these proteins encodes motifs of oligomer polydispersity, dissociation, and expansion to achieve functional diversity and regulation.
Keywords: Hsp27; chaperones; electron microscopy; multi-angle light scattering; native state mass spectrometry; proteostasis; sHSP; small heat-shock protein.
Copyright © 2018 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of Interests
The authors declare no competing interests.
Figures


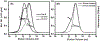
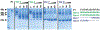
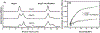
References
-
- Andley UP (2007). Crystallins in the eye: Function and pathology. Progress in Retinal and Eye Research 26, 78–98. - PubMed
-
- Benesch JL, Aquilina JA, Ruotolo BT, Sobott F, and Robinson CV (2006). Tandem mass spectrometry reveals the quaternary organization of macromolecular assemblies. Chem Biol 13, 597–605. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

