Arrestin-3 interaction with maternal embryonic leucine-zipper kinase
- PMID: 31352007
- PMCID: PMC6717526
- DOI: 10.1016/j.cellsig.2019.109366
Arrestin-3 interaction with maternal embryonic leucine-zipper kinase
Abstract
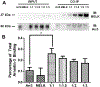
Maternal embryonic leucine-zipper kinase (MELK) overexpression impacts survival and proliferation of multiple cancer types, most notably glioblastomas and breast cancer. This makes MELK an attractive molecular target for cancer therapy. Yet the molecular mechanisms underlying the involvement of MELK in tumorigenic processes are unknown. MELK participates in numerous protein-protein interactions that affect cell cycle, proliferation, apoptosis, and embryonic development. Here we used both in vitro and in-cell assays to identify a direct interaction between MELK and arrestin-3. A part of this interaction involves the MELK kinase domain, and we further show that the interaction between the MELK kinase domain and arrestin-3 decreases the number of cells in S-phase, as compared to cells expressing the MELK kinase domain alone. Thus, we describe a new mechanism of regulation of MELK function, which may contribute to the control of cell fate.
Keywords: Arrestin; Cell fate signaling; Maternal embryonic leucine-zipper kinase (MELK); Protein-protein interactions.
Copyright © 2019 Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of Interest
The authors declare no conflict of interest.
Figures





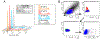
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

