Protection from Endotoxin Shock by Selective Targeting of Proinflammatory Signaling to the Nucleus Mediated by Importin Alpha 5
- PMID: 31533951
- PMCID: PMC6768080
- DOI: 10.4049/immunohorizons.1900064
Protection from Endotoxin Shock by Selective Targeting of Proinflammatory Signaling to the Nucleus Mediated by Importin Alpha 5
Abstract
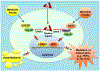
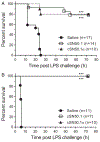
Endotoxin shock is induced by LPS, one of the most potent virulence factors of the Gram-negative bacteria that cause sepsis. It remains unknown if either proinflammatory stress-responsive transcription factors (SRTFs), ferried to nucleus by importin α5, or lipid-regulating sterol regulatory element binding proteins (SREBPs), transported to the nucleus by importin β1, mediate endotoxin shock. A novel cell-penetrating peptide targeting importin α5 while sparing importin β1 protected 80% of animals from death in response to a high dose of LPS. This peptide suppresses inflammatory mediators, liver glycogen depletion, endothelial injury, neutrophil trafficking, and apoptosis caused by LPS. In d-galactosamine-pretreated mice challenged by 700-times lower dose of LPS, rapid death through massive apoptosis and hemorrhagic necrosis of the liver was also averted by the importin α5-selective peptide. Thus, using a new tool for selective suppression of nuclear transport, we demonstrate that SRTFs, rather than SREBPs, mediate endotoxin shock.
Copyright © 2019 The Authors.
Conflict of interest statement
DISCLOSURES
Y.L., R.A.V., J.Z., and J.H. are coinventors of patents assigned to Vanderbilt University and the US Department of Veterans Affairs. The other authors have no financial conflicts of interest.
Figures
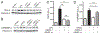
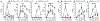

References
-
- Andreasen AS, Krabbe KS, Krogh-Madsen R, Taudorf S, Pedersen BK, and Møller K. 2008. Human endotoxemia as a model of systemic inflammation. Curr. Med. Chem 15: 1697–1705. - PubMed
-
- Opal SM 2010. New perspectives on immunomodulatory therapy for bacteraemia and sepsis. Int. J. Antimicrob. Agents 36(Suppl. 2): S70–S73. - PubMed
-
- Hawiger J 2001. Innate immunity and inflammation: a transcriptional paradigm. Immunol. Res 23: 99–109. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

