A conserved role of αA-crystallin in the development of the zebrafish embryonic lens
- PMID: 26149094
- PMCID: PMC4638411
- DOI: 10.1016/j.exer.2015.07.001
A conserved role of αA-crystallin in the development of the zebrafish embryonic lens
Abstract
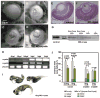
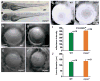
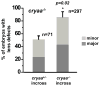
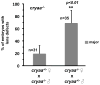
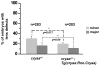
αA- and αB-crystallins are small heat shock proteins that bind thermodynamically destabilized proteins thereby inhibiting their aggregation. Highly expressed in the mammalian lens, the α-crystallins have been postulated to play a critical role in the maintenance of lens optical properties by sequestering age-damaged proteins prone to aggregation as well as through a multitude of roles in lens epithelial cells. Here, we have examined the role of α-crystallins in the development of the vertebrate zebrafish lens. For this purpose, we have carried out morpholino-mediated knockdown of αA-, αBa- and αBb-crystallin and characterized the gross morphology of the lens. We observed lens abnormalities, including increased reflectance intensity, as a consequence of the interference with expression of these proteins. These abnormalities were less frequent in transgenic zebrafish embryos expressing rat αA-crystallin suggesting a specific role of α-crystallins in embryonic lens development. To extend and confirm these findings, we generated an αA-crystallin knockout zebrafish line. A more consistent and severe lens phenotype was evident in maternal/zygotic αA-crystallin mutants compared to those observed by morpholino knockdown. The penetrance of the lens phenotype was reduced by transgenic expression of rat αA-crystallin and its severity was attenuated by maternal αA-crystallin expression. These findings demonstrate that the role of α-crystallins in lens development is conserved from mammals to zebrafish and set the stage for using the embryonic lens as a model system to test mechanistic aspects of α-crystallin chaperone activity and to develop strategies to fine-tune protein-protein interactions in aging and cataracts.
Keywords: Alpha-crystallin; Cataract; Chaperone; Lens development; Maternal transcript; Morpholino; Small heat shock protein; TALEN; Zebrafish.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures

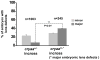
References
-
- Andley UP. Crystallins in the eye: Function and pathology. Progress in Retinal & Eye Researcmh. 2007;26:78–98. - PubMed
-
- Andley UP, Song Z, Wawrousek EF, Bassnett S. The molecular chaperone aA-crystallin enhances lens epithelial cell growth and resistance to UVA stress. Journal of Biological Chemistry. 1998;273:31252–31261. - PubMed
-
- Andley UP, Song Z, Wawrousek EF, Brady JP, Bassnett S, Fleming TP. Lens epithelial cells derived from aB-crystallin knockout mice demonstrate hyperproliferation and genomic instability. FASEB Journal. 2001;15:221–229. - PubMed
-
- Andley UP, Song Z, Wawrousek EF, Fleming TP, Bassnett S. Differential protective activity of aA- and aB-crystallin in lens epithelial cells. Journal of Biological Chemistry. 2000;275:36823–36831. - PubMed
-
- Bassnett S. Fiber cell denucleation in the primate lens. Investigative Ophthalmology & Visual Science. 1997;38:1678–1687. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

